Fluorescence-activated cell sorting (FACS) registered trademark of Becton Dickinson
To determine more specific biochemical
properties of a cell we can also apply flow cytometry. In order to do so, is
necessary to apply in the sample;
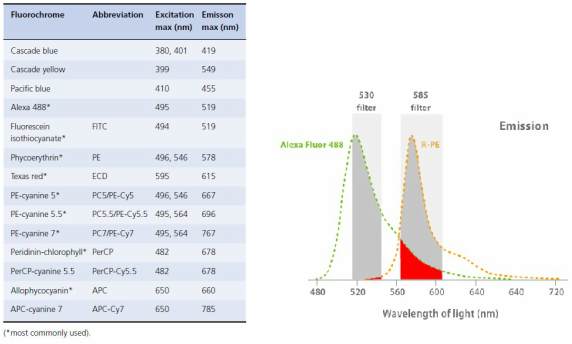
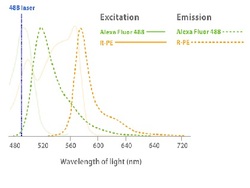
Some flow cytometers use only one laser, so we need to select flurecents that have the similar excitation peaks but emission peaks in separeted wavelenght of light (nm). If the wavelenghts are enought apart differencial data can be collected.
To optimize the fluorescent signals, optical filters specific to a wavelength range are placed in front of the photomultiplier, allowing only a narrow range of wavelength to reach the detector. For example 530 filter for Alexa Fluor 488 and 585 for R-PE.
- dyes that can bind directly to the cell;
- flurochromes that are bound to ligands, such as monoclonal antibodies;
Some flow cytometers use only one laser, so we need to select flurecents that have the similar excitation peaks but emission peaks in separeted wavelenght of light (nm). If the wavelenghts are enought apart differencial data can be collected.
To optimize the fluorescent signals, optical filters specific to a wavelength range are placed in front of the photomultiplier, allowing only a narrow range of wavelength to reach the detector. For example 530 filter for Alexa Fluor 488 and 585 for R-PE.
In red we have the Spectral Overlap: the emission spectra for a given fluorochrome can cover a range of wavelengths, allowing fluorescence spill over to a detector designed for a different fluorochrome.
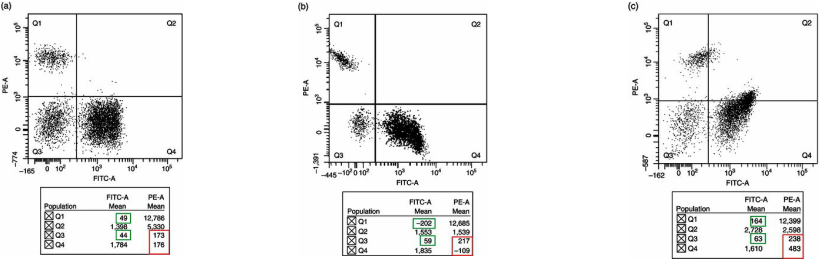
For accurate analysis of data, the spectral overlap between fluorochromes must be corrected. This compensation is done by correcting the inappropriate signals generated by the fluorochrome responsible for the overlap, with the use of compensation controls, manually and visually set.
However, when using multiple fluorochromes in modern flow cytometers this process is much more difficult to carry out manually. Therefore, current four and six colour flow cytometers usually have a software programme to aid with this potentially complex compensation set up process.
For accurate analysis of data, the spectral overlap between fluorochromes must be corrected. This compensation is done by correcting the inappropriate signals generated by the fluorochrome responsible for the overlap, with the use of compensation controls, manually and visually set.
However, when using multiple fluorochromes in modern flow cytometers this process is much more difficult to carry out manually. Therefore, current four and six colour flow cytometers usually have a software programme to aid with this potentially complex compensation set up process.
Fig. http://probes.invitrogen.com/resources/education/tutorials/4Intro_Flow/player.html
Leach, M., Drummond, M., and Doig, A. (2013) Pratical Flow Cytometry in Haematology Diagnosis. John Wiley & Sons, Ltd.
Reference:Leach, M., Drummond, M., and Doig, A. (2013) Pratical Flow Cytometry in Haematology Diagnosis. John Wiley & Sons, Ltd.
http://www.stemcell.umn.edu/prod/groups/med/@pub/@med/documents/asset/med_80691.pdf
Leach, M., Drummond, M., and Doig, A. (2013) Pratical Flow Cytometry in Haematology Diagnosis. John Wiley & Sons, Ltd.
Reference:Leach, M., Drummond, M., and Doig, A. (2013) Pratical Flow Cytometry in Haematology Diagnosis. John Wiley & Sons, Ltd.
http://www.stemcell.umn.edu/prod/groups/med/@pub/@med/documents/asset/med_80691.pdf